Efficient Gold Nanoparticle Conjugation for Protein Studies: Revolutionizing Biomedical Research
Unlock the full potential of your protein research with advanced **efficient gold nanoparticle conjugation techniques**. This comprehensive guide delves into the core methods, optimization strategies, and groundbreaking applications of **gold nanoparticles in protein research**, providing insights for achieving stable and functional **gold nanoparticle protein conjugates** essential for cutting-edge biomedical applications. From **protein labeling with gold nanoparticles** to their role in **targeted drug delivery**, understand how these nanoscale marvels are transforming the landscape of biological studies.
Explore Our Gold Nanoparticle Conjugation Optimization Kit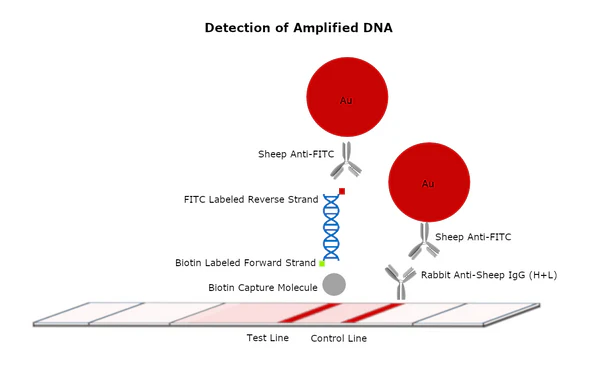
The Pivotal Role of Gold Nanoparticles in Protein Research
**Gold nanoparticles (AuNPs)** have emerged as indispensable tools across various scientific disciplines, particularly in biology and medicine. Their unique optical, electronic, and catalytic properties, combined with excellent biocompatibility and ease of surface functionalization, make them ideal candidates for a myriad of **application of gold nanoparticles in biology**. When it comes to **protein studies using gold nanoparticles**, the ability to precisely attach proteins to their surface is paramount. This process, known as **gold nanoparticle conjugation techniques**, enables researchers to harness the combined power of nanoscale materials and biological specificity.
The demand for **efficient conjugation methods for proteins** stems from the need for highly stable, specific, and functionally active **gold nanoparticle protein conjugates**. These conjugates are critical for advancing fields such as diagnostics, therapeutics, and fundamental biological inquiry. From developing ultra-sensitive biosensors to enabling precise cellular imaging and targeted drug delivery systems, the success largely hinges on the quality and efficiency of the conjugation process.
Understanding Gold Nanoparticle Conjugation Techniques
Achieving stable and functional **gold nanoparticle protein conjugates** requires a deep understanding of various **gold nanoparticle conjugation protocols**. The choice of method often depends on the specific protein, the desired application, and the characteristics of the gold nanoparticles themselves (e.g., size, surface chemistry). Here are some of the major **efficient conjugation methods for proteins**:
1. Passive Adsorption (Electrostatic Interaction)
This is one of the simplest methods, relying on the electrostatic attraction between negatively charged citrate-stabilized gold nanoparticles and positively charged regions of proteins. Proteins adsorb onto the gold surface, forming a protein corona. While straightforward, it can lead to non-specific binding and potential denaturation of the protein. Optimization involves careful control of pH and ionic strength to ensure stable **protein binding studies with gold nanoparticles**.
2. Covalent Conjugation
Covalent methods form strong, irreversible bonds between the protein and the gold nanoparticle surface, leading to highly stable conjugates. This is often preferred for applications requiring long-term stability in complex biological environments.
- Carbodiimide Chemistry (EDC/NHS): This widely used method involves activating carboxyl groups on the gold nanoparticle surface (or a linker molecule attached to it) with N-(3-Dimethylaminopropyl)-N′-ethylcarbodiimide hydrochloride (EDC) and N-hydroxysuccinimide (NHS). The activated carboxyl groups then react with primary amine groups of the protein, forming stable amide bonds. This is a robust approach for **functionalization of gold nanoparticles** for protein attachment.
- Thiol Chemistry: Gold has a strong affinity for sulfur. Proteins containing cysteine residues (with free thiol groups) can directly bind to the gold surface via Au-S bonds. Alternatively, the gold surface can be functionalized with thiol-terminated linkers (e.g., thiolated polyethylene glycol, PEG) which then react with proteins. This method is highly specific and provides excellent stability, making it popular for **protein labeling with gold nanoparticles**.
- Click Chemistry: This bioorthogonal reaction involves the rapid and specific formation of a bond between two functional groups (e.g., azide and alkyne). By functionalizing gold nanoparticles with one group and the protein with the other, highly efficient and specific conjugation can be achieved under mild conditions, minimizing protein denaturation. This represents an **innovative uses of gold nanoparticles in research** for precise conjugation.
3. Ligand Exchange
In this method, existing ligands on the gold nanoparticle surface (e.g., citrate) are replaced by molecules with a stronger affinity for gold, such as thiols, amines, or phosphines. These new ligands can be designed to directly bind to proteins or to present reactive groups for subsequent covalent attachment. This allows for precise control over the surface chemistry and is crucial for advanced **gold nanoparticle synthesis for proteins** with specific functionalities.
4. Streptavidin-Biotin System
This highly specific and robust biological interaction is widely exploited. Gold nanoparticles are functionalized with streptavidin, which then binds with high affinity to biotinylated proteins. This method offers excellent control and minimal non-specific binding, making it ideal for sensitive diagnostic assays and **high-throughput gold nanoparticle conjugation**.
Optimizing Gold Nanoparticle Conjugation for Peak Performance
**Nanoparticle conjugation optimization** is crucial for maximizing protein loading, preserving protein activity, and ensuring the stability of **gold nanoparticle protein conjugates**. Several factors must be carefully considered:
- pH Control: The pH of the conjugation buffer significantly impacts protein charge, solubility, and stability, as well as the surface charge of the gold nanoparticles. Optimal pH ensures maximum electrostatic interaction (for passive adsorption) or optimal reactivity for covalent methods.
- Protein Concentration: An adequate protein concentration is needed to achieve desired loading without causing aggregation. Too little protein can lead to insufficient coverage, while too much can lead to aggregation or non-specific binding.
- Incubation Time and Temperature: These parameters influence the kinetics of the binding reaction. Optimal time ensures complete reaction without prolonged exposure that could lead to protein degradation or aggregation.
- Blocking Agents: After conjugation, it's often necessary to block any remaining unoccupied binding sites on the gold nanoparticle surface to prevent non-specific adsorption of other proteins from biological samples. Common blocking agents include BSA, PEG, or casein. This is vital for maintaining **gold nanoparticle stability in biological systems**.
- Purification and Characterization: Post-conjugation purification removes unbound proteins and reagents. Subsequent **characterization of gold nanoparticle conjugates** (e.g., UV-Vis spectroscopy, DLS, TEM, zeta potential, SDS-PAGE) is essential to confirm successful conjugation, assess stability, and quantify protein loading.
Recent Major Applications of Gold Nanoparticles in Protein Studies
The versatility of **gold nanoparticles in protein research** has led to their widespread adoption across numerous biomedical applications. Here are some key areas where **gold nanoparticle protein conjugates** are making a significant impact:
1. Advanced Biosensors and Diagnostics
Gold nanoparticles are at the forefront of developing highly sensitive and rapid diagnostic tools. By conjugating specific antibodies or aptamers (protein-binding nucleic acids) to AuNPs, researchers can create biosensors capable of detecting target proteins, biomarkers, or pathogens with unparalleled sensitivity. For example, in lateral flow assays (like rapid COVID-19 tests), gold nanoparticles conjugated with antibodies produce the visible colored line, indicating the presence of the target antigen. The **advantages of gold nanoparticles in protein studies** for diagnostics include their strong optical properties (colorimetric detection) and high surface area for multi-analyte detection.
2. Targeted Drug Delivery Systems
**Gold nanoparticles for targeted drug delivery** represent a promising avenue for treating diseases like cancer. Proteins such as antibodies, growth factors, or peptides can be conjugated to AuNPs, enabling them to specifically recognize and bind to receptors overexpressed on cancer cells. This targeted delivery minimizes systemic toxicity of chemotherapeutic drugs, improving therapeutic efficacy. An example includes AuNPs conjugated with HER2 antibodies to deliver drugs specifically to HER2-positive breast cancer cells, significantly reducing off-target effects.
3. Cellular Imaging and Bioimaging
The excellent optical properties of gold nanoparticles, particularly their surface plasmon resonance, make them ideal for **gold nanoparticles for imaging proteins** and cellular structures. Proteins conjugated to AuNPs can act as probes to visualize specific proteins or processes within living cells with high resolution and contrast. Techniques like dark-field microscopy, surface-enhanced Raman scattering (SERS), and two-photon luminescence utilize AuNPs for deep tissue imaging and real-time tracking of biological events. For instance, AuNPs conjugated with fluorescent proteins or antibodies are used to track protein trafficking or receptor dimerization on cell membranes.
4. Protein-Protein Interaction Studies
**Gold nanoparticles and protein interactions** are invaluable for understanding complex biological pathways. By conjugating one protein to AuNPs and observing its interaction with another protein in solution or on a surface, researchers can gain insights into binding kinetics, affinity, and specificity. Techniques like localized surface plasmon resonance (LSPR) sensing, which measures shifts in the AuNP's optical properties upon protein binding, are powerful tools for **protein binding studies with gold nanoparticles**, providing real-time, label-free detection of interactions relevant to disease mechanisms.
5. Gene and Protein Delivery
Beyond drug delivery, gold nanoparticles can also facilitate the intracellular delivery of genetic material (DNA, RNA) or therapeutic proteins. By conjugating specific proteins (e.g., cell-penetrating peptides) to AuNPs, the nanoparticles can effectively overcome cellular barriers, delivering their cargo directly into the cytoplasm or nucleus. This holds immense potential for gene therapy and protein replacement therapies.
Challenges and Future Directions in Gold Nanoparticle Conjugation
Despite the immense **advantages of gold nanoparticles in protein studies**, challenges remain. Ensuring the long-term **gold nanoparticle stability in biological systems** is critical, as proteins can desorb or denature over time. Non-specific binding in complex biological matrices can also lead to false positives in diagnostic assays or off-target effects in therapeutic applications. Reproducibility of **gold nanoparticle conjugation protocols** across different batches and laboratories is another area requiring standardization.
Future directions in **nanoparticle conjugation optimization** focus on developing novel surface chemistries that offer even greater specificity, stability, and biocompatibility. The integration of artificial intelligence and machine learning for predicting optimal conjugation conditions based on protein characteristics is also emerging. Furthermore, advancements in **high-throughput gold nanoparticle conjugation** methods will accelerate the discovery and development of new diagnostic and therapeutic platforms, pushing the boundaries of **innovative uses of gold nanoparticles in research**.
Optimize Your Research with Our Gold Nanoparticle Conjugation Kit
Ready to streamline your **gold nanoparticle conjugation techniques** and achieve superior results in your **protein studies using gold nanoparticles**? Our Gold Nanoparticle Conjugation Optimization Kit is designed to provide everything you need for efficient, stable, and reproducible **gold nanoparticle protein conjugates**. Enhance your **nanoparticle conjugation optimization** process today!
Learn More About Our Optimization KitFrequently Asked Questions About Gold Nanoparticle Conjugation
Have Questions? Contact Us!
Our experts are here to assist you with your gold nanoparticle conjugation needs.
