Introduction to Ammonium-Based Ionic Liquids with Gold NanoUrchins
Dive into the cutting-edge intersection of materials science and green chemistry. This article explores the fascinating world of ammonium-based ionic liquids and their transformative role in the precise synthesis and diverse applications of gold nanourchins. Discover how these innovative materials are shaping the future of sustainable nanotechnology, from advanced catalysis to targeted drug delivery.
Explore Gold NanoUrchins Kit
The Dawn of Green Solvents: Understanding Ammonium-Based Ionic Liquids
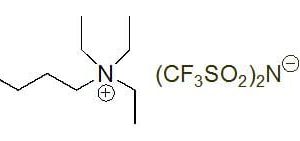
In the quest for more sustainable and efficient chemical processes, ionic liquids have emerged as revolutionary solvents. Among these, ammonium-based ionic liquids stand out due to their distinct properties and versatility. Unlike traditional volatile organic solvents (VOCs), ionic liquids are salts that are liquid at or near room temperature. This fundamental difference bestows upon them exceptional characteristics, including negligible vapor pressure, non-flammability, high thermal stability, and remarkable tunability in their physical and chemical properties.
The unique structure of ammonium-based solvents, comprising an organic ammonium cation and various anions, allows for precise tailoring of their characteristics. This tunability is crucial for specific applications, making them ideal candidates for a wide array of industrial and research purposes. Their contribution to green chemistry ionic liquids is profound, as they significantly reduce the environmental footprint of chemical syntheses by minimizing hazardous waste and enabling cleaner processes. The inherent non-volatility of these solvents means less air pollution and safer working environments, aligning perfectly with the principles of sustainable chemistry.
Furthermore, the processes for ionic liquid purification have advanced, ensuring high purity products essential for sensitive applications. While discussions around ammonium ionic liquid safety are ongoing, continuous research and development are focused on designing inherently safer ionic liquid formulations and establishing best practices for their handling and disposal. The numerous ammonium-based ionic liquid benefits underscore their potential to replace conventional solvents in many high-value applications, paving the way for a more environmentally conscious chemical industry.
Unveiling Gold NanoUrchins: Structure, Synthesis, and Extraordinary Properties
Gold nanourchins represent a fascinating class of noble metal nanostructures, distinguished by their characteristic spiky, anisotropic morphology, resembling the sea urchin. These intricate structures are composed of a central gold nanoparticle core from which numerous sharp, crystalline spikes protrude. This unique morphology is not merely aesthetically pleasing; it is critical to their enhanced properties, especially their significantly increased surface area and localized surface plasmon resonance (LSPR) properties, which are far superior to those of spherical gold nanoparticles.
The controlled gold nanourchins synthesis is a complex yet crucial step in harnessing their potential. Traditional synthesis methods often involve harsh reducing agents and stabilizing agents that can leave unwanted residues. This is where ionic liquids, particularly ammonium-based ones, play a transformative role. They act as both solvents and templating agents, offering an unparalleled medium for the precise control over the nucleation and growth of these intricate gold nanostructures. The unique environment provided by ammonium ionic liquids facilitates anisotropic growth, leading to the formation of the distinctive spikes and ensuring superior nanoparticle stabilization during and after synthesis. This approach aligns perfectly with the goals of nanotechnology and ionic liquids research, aiming for cleaner, more efficient, and more controllable synthesis routes.
The extraordinary properties of gold nanourchins stem directly from their morphology. Their sharp tips act as "hot spots" for electromagnetic field enhancement, making them exceptionally effective in applications requiring high sensitivity, such as surface-enhanced Raman spectroscopy (SERS). Their enhanced light absorption and scattering properties also make them excellent candidates for photothermal therapies and advanced imaging techniques. The ability to precisely tune their optical properties by controlling their size and spike density through tailored ionic liquid formulations opens up vast possibilities for their application across various scientific and technological domains.
The Synergistic Power: Ammonium Ionic Liquids in Gold NanoUrchin Synthesis and Beyond
The synergy between ammonium ionic liquids and gold nanourchins synthesis is a prime example of how innovative solvent design can revolutionize nanomaterial fabrication. Ionic liquids provide a unique reaction environment that is highly conducive to the controlled growth of anisotropic nanostructures. Their high polarity, tunable viscosity, and ability to act as soft templates or even capping agents allow for exquisite control over the shape and size of the resulting gold nanourchins.
In traditional aqueous synthesis, achieving uniform and stable spiky morphologies can be challenging due to rapid, uncontrolled growth and aggregation. However, the use of ammonium-based solvents mitigates these issues. They can slow down the reduction kinetics of gold precursors, allowing for a more gradual and directed growth of the spikes. Furthermore, the ionic nature of these liquids can influence the facet-specific growth of gold crystals, promoting the formation of sharp edges and tips that define the urchin-like structure. This level of control is paramount for producing high-quality gold nanostructures with consistent properties, essential for reliable performance in advanced applications.
Beyond synthesis, ionic liquid properties also contribute to the long-term stability and dispersibility of gold nanourchins. They can act as excellent dispersants, preventing aggregation and maintaining the integrity of the nanostructures in various media. This enhanced stability is crucial for their integration into complex systems, such as biological environments for drug delivery or catalytic reactors. The ongoing ionic liquid research in this area is constantly uncovering new ways to leverage these solvent properties for novel nanomaterial designs and improved performance, pushing the boundaries of nanotechnology and ionic liquids.
Recent Major Applications: Where Ammonium Ionic Liquids and Gold NanoUrchins Shine
The combined prowess of ammonium ionic liquids and gold nanourchins has opened doors to groundbreaking advancements across numerous fields. Their unique properties make them invaluable assets in addressing some of the most pressing challenges in science and technology.
1. Catalysis: Elevating Reaction Efficiency
One of the most significant ionic liquid applications is in catalysis, particularly when paired with gold nanourchins. The high surface area and numerous "hot spots" on gold nanourchins make them exceptionally active catalysts. They have shown remarkable efficiency in various organic reactions, including oxidation, reduction, and cross-coupling reactions. For instance, gold nanourchins in catalysis are being explored for the efficient degradation of environmental pollutants, selective organic transformations, and even in fuel cell applications. The ionic liquid medium not only facilitates the synthesis of these highly active catalysts but can also serve as a reaction medium itself, often enabling greener and more selective catalytic processes. This synergy promotes sustainable chemistry by reducing catalyst loading and improving reaction yields.
2. Electrochemistry: Advanced Sensors and Energy Storage
The field of electrochemistry heavily benefits from both ionic liquid properties and the unique features of gold nanoparticles. Ionic liquids in electrochemistry provide stable, wide electrochemical windows and high ionic conductivity, making them excellent electrolytes. When integrated with gold nanourchins, these systems form highly sensitive electrochemical sensors. The spiky morphology of nanourchins significantly increases the electroactive surface area, leading to enhanced detection limits for analytes, from biomarkers to environmental toxins. Furthermore, their application extends to advanced energy storage devices, such as supercapacitors and batteries, where the high surface area of the gold nanostructures combined with the efficient ion transport of ionic liquids can lead to improved energy density and cycling stability.
3. Drug Delivery and Biomedicine: Precision Therapeutics
In the biomedical arena, gold nanourchins for drug delivery represent a promising frontier. Their biocompatibility, tunable optical properties, and high surface area for drug loading make them ideal nanocarriers. They can be engineered to target specific cells or tissues, releasing therapeutic agents precisely where needed, thereby minimizing systemic side effects. The role of ionic liquids in pharmaceuticals is also gaining traction, particularly in drug formulation and solubility enhancement. While direct injection of ionic liquids is still under research, their use in synthesizing and stabilizing these crucial nanocarriers ensures the production of highly stable and functional gold nanourchins suitable for biological applications. This integration of nanotechnology and ionic liquids is paving the way for next-generation diagnostics and therapeutics.
4. Environmental Remediation: Cleaner Water and Air
Beyond the aforementioned, ionic liquid applications extend to environmental remediation. Gold nanourchins, often synthesized using green methods facilitated by ionic liquids, are being investigated for their ability to adsorb and degrade pollutants from water and air. Their catalytic activity can break down persistent organic pollutants, while their large surface area makes them effective adsorbents for heavy metals and other contaminants. This directly contributes to sustainable chemistry efforts to create a cleaner planet.
5. Biosensing and Diagnostics: Enhanced Sensitivity
The exceptional LSPR properties of gold nanourchins, amplified by their spiky structure, make them superb platforms for biosensing. They enable highly sensitive and selective detection of biomolecules, pathogens, and disease markers. The stability and functionalizability offered by ammonium-based ionic liquids in their synthesis ensure that these biosensors are robust and reliable for real-world diagnostic applications.
Future Outlook and Ongoing Ionic Liquid Research
The journey of ammonium-based ionic liquids and gold nanourchins is still in its early stages, yet the potential is immense. Ongoing ionic liquid research is focused on developing novel ionic liquid formulations with even more tailored properties, enhancing their stability, reducing their cost, and ensuring their long-term environmental safety. Researchers are exploring new synthesis pathways for gold nanourchins that are even more energy-efficient and scalable, leveraging the unique solvent properties of ionic liquids.
Challenges remain, particularly in scaling up the production of these advanced materials for industrial applications and fully understanding the long-term environmental impact and ammonium ionic liquid safety profiles. However, the continuous advancements in ionic liquid purification techniques and a deeper understanding of their interactions at the nanoscale are rapidly addressing these concerns. The convergence of nanotechnology and ionic liquids promises a future where materials are not only highly functional but also produced and utilized in a manner that is inherently sustainable and environmentally responsible. The benefits of ammonium-based ionic liquid benefits will continue to drive innovation in this exciting field, leading to breakthroughs in diverse sectors from energy to medicine.
Ready to Explore the Future of Nanotechnology?
Discover our innovative solutions and get started with your own research into reactant-free gold nanourchins.
Get Your Introduction Kit TodayFrequently Asked Questions
Ammonium-based ionic liquids are considered "green" solvents primarily because of their negligible vapor pressure, which eliminates the emission of volatile organic compounds (VOCs) into the atmosphere. Unlike traditional solvents, they are non-flammable and possess high thermal stability, contributing to safer chemical processes. Their tunability also allows for more selective reactions, reducing by-products and waste, aligning perfectly with the principles of sustainable chemistry and minimizing environmental impact.
Ionic liquids play a crucial role in gold nanourchins synthesis by acting as both a solvent and a soft template. Their unique properties, such as high polarity and tunable viscosity, allow for precise control over the nucleation and anisotropic growth of gold nanoparticles, leading to the distinctive spiky morphology. They help in nanoparticle stabilization, preventing aggregation and ensuring uniform distribution, which is vital for creating high-quality gold nanostructures with desired properties. This method contributes to cleaner and more controlled synthesis of gold nanoparticles.
Gold nanourchins, especially when synthesized using advanced methods involving ammonium ionic liquids, have diverse and impactful applications. Key areas include gold nanourchins in catalysis for efficient chemical reactions and pollutant degradation, advanced biosensing due to their enhanced LSPR properties, and promising roles in biomedical fields like gold nanourchins for drug delivery and photothermal therapy. Their unique morphology and amplified surface area make them superior to conventional spherical gold nanoparticles in many contexts, driving innovation in nanotechnology and ionic liquids.
The ammonium ionic liquid safety profile is a subject of ongoing research. While they offer significant advantages over traditional solvents (e.g., non-flammability, low volatility), their biocompatibility and environmental impact vary depending on their specific chemical structure (cation and anion combination). Many ammonium-based ionic liquid benefits are being explored for various applications, including some in ionic liquids in pharmaceuticals. However, comprehensive toxicological and ecotoxicological studies are continuously being conducted to ensure their safe and responsible use across all intended applications, emphasizing the need for careful selection and handling of specific ionic liquid formulations.
Hiyka is at the forefront of providing innovative solutions in this field, including reactant-free gold nanourchins and related materials. Our commitment to sustainable chemistry and advanced ionic liquid research aims to make these cutting-edge materials accessible for research and development. We focus on developing and supplying high-quality products that facilitate breakthroughs in ionic liquid applications, from novel synthesis methods to advanced functional materials for various industries. Our offerings are designed to support researchers and industries in harnessing the full potential of nanotechnology and ionic liquids.
Get in Touch
Have questions or need more information? Contact our experts today.