Welcome to the cutting edge of materials science, where the unique characteristics of ionic liquids converge with the remarkable properties of gold nanoparticles. This powerful combination is unlocking unprecedented possibilities across diverse fields, from sustainable chemistry to advanced medicine. Delve into the fascinating world of these innovative materials, exploring their fundamental properties, synthesis techniques, and the transformative ionic liquids applications that are redefining what's possible in modern technology and research.

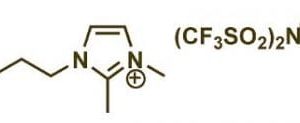
Ionic liquids (ILs) are molten salts with melting points below 100 °C, often even at room temperature. Unlike conventional molecular solvents, ILs are composed entirely of ions, giving them a distinct set of ionic liquids properties that make them exceptionally versatile. Key attributes include negligible vapor pressure, high thermal stability, non-flammability, excellent ionic conductivity, and a remarkable ability to dissolve a wide range of organic and inorganic compounds. Their tunability is perhaps their most celebrated feature; by selecting different cations and anions, researchers can tailor their physical and chemical properties to suit specific applications, earning them the moniker "designer solvents." This inherent flexibility positions them as prime candidates for advancing ionic liquids and green chemistry initiatives, offering sustainable alternatives to volatile organic compounds (VOCs).
The absence of volatility means no harmful emissions, contributing significantly to a safer and cleaner environment. Furthermore, their high thermal stability allows for reactions at elevated temperatures without solvent evaporation, and their distinct solvent properties often lead to enhanced reaction rates and selectivities. This unique combination of features makes them invaluable across various scientific and industrial sectors, paving the way for novel processes and products.
Gold nanoparticles (AuNPs) are nanoscale particles of gold, typically ranging from 1 to 100 nanometers in diameter. At this scale, gold exhibits fascinating quantum mechanical properties that differ significantly from bulk gold. These include unique optical properties (leading to vibrant colors), excellent biocompatibility, and high surface-to-volume ratios, making them ideal for surface-dependent reactions. Their localized surface plasmon resonance (LSPR) makes them highly sensitive to their environment, a property exploited in various sensing applications. The stability of gold nanoparticles is crucial for their long-term performance, and their surface can be easily modified with various ligands, a process known as gold nanoparticles surface modification, to impart specific functionalities or improve their dispersion in different media.
The development of precise gold nanoparticles synthesis techniques is vital for controlling their size, shape, and distribution, which directly impacts their properties and performance in diverse applications. Common methods include chemical reduction, electrochemical synthesis, and green synthesis routes. These tiny structures are at the forefront of ionic liquids and nanotechnology research, offering unparalleled opportunities to innovate in fields previously limited by conventional materials.
The integration of gold nanoparticles in ionic liquids represents a powerful synergy. Ionic liquids provide an exceptional medium for the synthesis, stabilization, and application of AuNPs. Their high polarity and ability to act as both solvent and template can lead to better control over particle size and morphology during synthesis of ionic liquids containing gold nanoparticles. Moreover, the non-volatile nature of ILs minimizes particle aggregation, enhancing the stability of gold nanoparticles over extended periods, which is a critical challenge in nanoparticle research.
The unique solvent properties of ILs allow for the dissolution of various gold precursors and reducing agents, facilitating cleaner and more efficient synthesis processes. Furthermore, the ionic environment can passivate the surface of AuNPs, preventing their agglomeration and maintaining their nanoscale properties. This combination is particularly attractive for developing robust and reusable nanohybrid systems.
The convergence of ionic liquids and gold nanoparticles has opened doors to groundbreaking ionic liquids applications across numerous high-impact sectors. Here are some of the most significant advancements:
One of the most prominent ionic liquids applications in catalysis involves their use as reaction media and supports for AuNPs. Gold nanoparticles are excellent catalysts for a variety of reactions, including oxidation, reduction, and coupling reactions. When dispersed in ionic liquids, their catalytic activity can be enhanced, and separation/recycling becomes significantly easier. For instance, AuNPs in ILs have shown remarkable efficiency in the oxidation of CO to CO2 at room temperature, a critical reaction for air purification and fuel cell applications. The ILs prevent leaching of the gold catalyst, ensuring high stability and reusability, aligning perfectly with principles of ionic liquids and green chemistry.
The high ionic conductivity and wide electrochemical windows of ILs make them ideal electrolytes for energy devices. When combined with gold nanoparticles, their potential in ionic liquids for energy storage is amplified. AuNPs can act as electrode modifiers, enhancing charge transfer kinetics and increasing the surface area for electrochemical reactions in supercapacitors and lithium-ion batteries. For example, AuNP-modified electrodes in IL electrolytes have demonstrated improved performance in dye-sensitized solar cells and fuel cells, offering pathways to more efficient and durable energy solutions.
The biocompatibility of gold nanoparticles makes them excellent candidates for biomedical applications, including gold nanoparticles for drug delivery and diagnostics. While ILs themselves have emerging roles in pharmaceuticals, their combination with AuNPs can create novel platforms. For instance, AuNPs synthesized or stabilized in specific ionic liquid solvents can be designed for targeted drug delivery, allowing therapeutic agents to reach specific cells or tissues with reduced side effects. Research also explores their use in biosensing and bioimaging, leveraging the unique optical properties of AuNPs in biocompatible IL matrices.
The non-volatile nature and tunability of ILs, coupled with the catalytic prowess of AuNPs, make this duo powerful for environmental applications. Ionic liquids for CO2 capture is a rapidly developing area, where ILs can selectively absorb CO2. The incorporation of AuNPs can potentially enhance the efficiency of CO2 conversion or detection. Furthermore, ionic liquid extraction methods benefit from the presence of AuNPs, which can act as sorbents or catalysts for the removal of pollutants from water or industrial streams, offering sustainable approaches to environmental challenges and minimizing the environmental impact of ionic liquids themselves by enabling their efficient recycling.
In ionic liquids in materials science, the combination facilitates the synthesis of novel nanomaterials with tailored properties. AuNPs can serve as building blocks or nucleation sites within IL matrices for creating complex nanostructures. In ionic liquids in electrochemistry, the high conductivity and stability of ILs, coupled with the electrocatalytic activity of AuNPs, are being explored for advanced sensors, electroplating, and electrosynthesis. The precise characterization of ionic liquids and the resulting nanostructures is crucial for optimizing these applications.
Despite their immense potential, challenges remain. Ensuring the long-term stability of gold nanoparticles in various IL environments, particularly under harsh reaction conditions, is an ongoing research focus. While ILs are often touted as "green," a thorough assessment of the environmental impact of ionic liquids is still necessary, particularly concerning their biodegradability and toxicity profiles for large-scale industrial applications. Research into more cost-effective and scalable synthesis of ionic liquids and their integration with AuNPs is also crucial for broader adoption.
The future of ionic liquids and nanotechnology is bright, with continuous innovation in designing novel ILs and advanced gold nanoparticles synthesis techniques. Expect to see further breakthroughs in their use for advanced sensing, efficient energy conversion, and smart materials, solidifying their role in shaping a sustainable technological future.
Explore our range of high-quality ionic liquids designed for diverse research and industrial applications:








Have questions or need more information about ionic liquids or gold nanoparticles? Reach out to our experts.